Allplex™ STI/BV Panel Assays is a multiplex real-time PCR assay developed using the proprietary MuDT™ technology. This assay detects and identifies 4 viruses, 16 bacteria, 1 parasite and 7 candida spp. related to sexually transmitted infections, and provides multiple quantification information of targets with individual Ct value in a single channel.
Key Features and Benefits
-
Full coverage of causative pathogens
Comprehensive assay for the detection and identification of 28 STI related pathogens
-

Multiplex real-time PCR
Simultaneous detection of 4 viruses, 16 bacteria, 1 parasite and 7 candida spp. related to sexually transmitted infections
-

Whole process validation
Whole process validation from extraction to PCR by whole process control
-
User-friendly workflow
Convenient workflow using Seegene’s automated one platform
-

Efficient syndromic test
The syndromic screening test for efficient and cost-effectiveness patient care
-

Proper patient care
Accurate test results allow to quick and proper treatment
-

Multi-Ct in a single channel
Individual Ct value of multiple analytes in a single channel of real-time PCR instrument (MuDT™ Technology)
-

Automatic data analyzer
Automated data interpretation and LIS interlocking with Seegene Viewer
-

Proper patient care
Quick and proper treatment provided by accurate test results
-

Informative assay
Assistant of appropriate treatment and management for co-infection
-

UDG system
Utilization of the UDG system to prevent carry-over contamination
-
Analytes
Allplex™
Bacterial Vaginosis plus Assay -
Allplex™
Candidiasis Assay- Candida albicans (CA)
- Candida dubliniensis (CD)
- Candida glabrata (CG)
- Candida krusei (CK)
- Candida lusitaniae (CL)
- Candida parapsilosis (CP)
- Candida tropicalis (CTp)
- Internal Control (IC)
-
Allplex™
Genital ulcer Assay- Cytomegalovirus (CMV)
- Haemophilus ducreyi (HD)
- Herpes simplex virus type 1 (HSV1)
- Herpes simplex virus type 2 (HSV2)
- Lymphogranuloma venereum (LGV)
- Treponema pallidum (TP)
- Varicella-zoster virus (VZV)
- Internal Control (IC)
-
Allplex™
STI Essential Assay- Chlamydia trachomatis (CT)
- Mycoplasma genitalium (MG)
- Mycoplasma hominis (MH)
- Neisseria gonorrhoeae (NG)
- Trichomonas vaginalis (TV)
- Ureaplasma parvum (UP)
- Ureaplasma urealyticum (UU)
- Internal Control (IC)
-
Specimens
- Genital swab
- Urine
- Liquid based cytology (e.g., ThinPrep® and Surepath™)
- Oropharyngeal swab(for STI Essential Assay only)
- Ano-rectal(for STI Essential Assay only)
-
Ordering Information
ProductCat No. / SizeAllplex™ Bacterial Vaginosis plus AssaySD10320Z / 25 rxns [3]
SD10159X / 100 rxnsAllplex™ Candidiasis AssayAllplex™ Genital ulcer AssayAllplex™ STI Essential Assay
Result
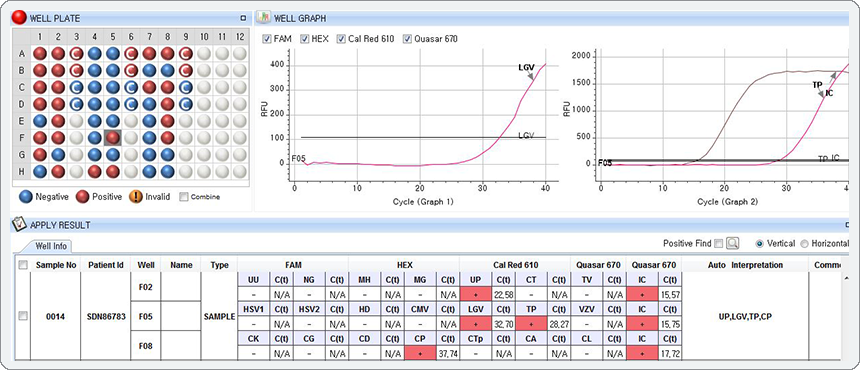
Multiple Ct value in a single channel
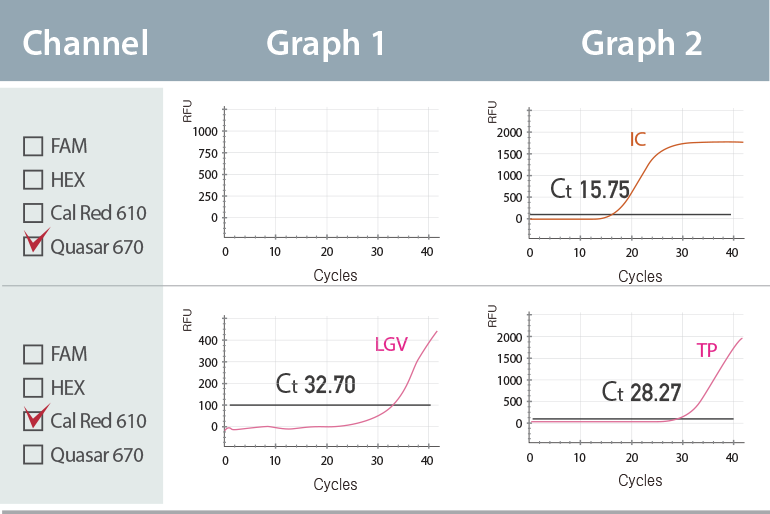
The result represents the co-infection of Lymphogranuloma venereum (LGV), Treponema pallidum (TP) in Cal Red 610 channel with Ct values of 32.70 (Graph 1) and 28.27 (Graph 2), respectively.
